The most extreme example known is the asteroid named 33 Polyhymnia, which is located in the main belt between Mars and Jupiter its density has been calculated as about 75 g/cm 3. Objects-typically, astronomical bodies-with densities higher than that are considered "compact ultradense objects" or CUDOs. 
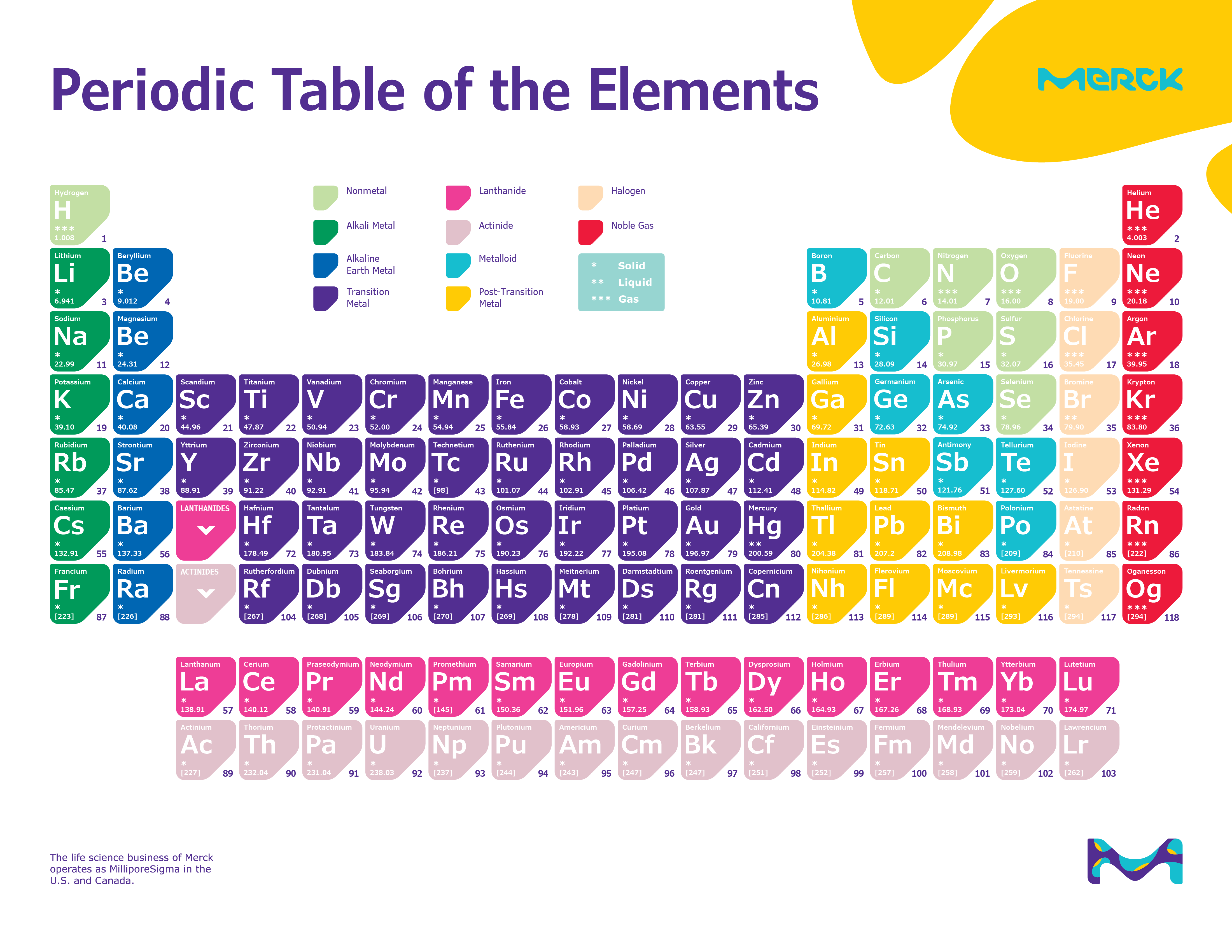
The densest stable element is the rare platinoid metal osmium (Z=76) its density of 22.59 g/cm 3 is about twice that of lead. And as, in general, the density of elements tends to rise with their atomic mass, these superheavy elements can be expected to be extremely dense. In particular, an "island of nuclear stability" is predicted at about Z=164. Those with atomic numbers between 105 and 118 have been made experimentally but are radioactive and unstable with very short half-lives and, therefore, are only of academic and research interest.Įlements with Z>118 have not yet been observed, but properties have been predicted for some of them. 
Superheavy elements are defined as those with a very high number of protons (high atomic number), generally considered to be those with Z>104. This work has now been published in The European Physical Journal Plus. They modeled the properties of such elements using the Thomas-Fermi model of atomic structure, concentrating particularly on a proposed "island of nuclear stability" at and around Z=164 and extending their method further to include more exotic types of ultra-dense material. Chemistry LibreTexts.Jan Rafelski and his team at the Department of Physics, The University of Arizona, Tucson, U.S., suggest that this could consist of superheavy elements with atomic number (Z) higher than the limit of the current periodic table. Atomic Radius Trend in Periodic Table (Simple Explanation). These factors are interdependent and determine the overall size of an atom. The factors affecting atomic radius are nuclear charge, electron configuration, and distance of the valence electrons from the nucleus. Experimental methods, such as X-ray crystallography, electron diffraction, and spectroscopy, are used to measure atomic radius. It helps us understand how atoms interact and affect their chemical properties. By analyzing the wavelengths of light that are absorbed or emitted by an atom, scientists can determine its atomic radius.Ītomic radius is the size of an atom, measured in picometers, from the center of the nucleus to the outermost electron in the electron cloud.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
